Lash Lure
In October, 1933, Paramount released a newsreel about the proposed changes to the 1906 Food and Drug Act which included footage describing how several women had suffered corneal damaged from using an ‘eyelash beautifier’. The ‘eyelash beautifier’ was not mentioned by name and many assumed that any eyelash product was suspect. Sales of mascara plummeted.
The unnamed product was an eyelash and eyebrow dye called Lash Lure that was manufactured by the Cosmetic Manufacturing Company of Los Angeles, California. It contained paraphenylenediamine (PPD), an aniline (coal tar) dye widely used in the hairdressing industry – now considered a potent allergen.
Lash lure was not the only eyelash and eyebrow dye on the American market at the time: others included Di-Lash, Coloura, Ey-Tec, Ey-dolize, Perma Coleur and Larieuse. Many of these contained PPD, however, Lash Lure was the only one involved in a case where a woman had become totally blind. In this case, the woman’s eyebrows had been plucked before being dyed which allowed them to become infected with Staphylococcus aureus (Golden Staph). The infection spread to her eyes, swollen with the allergic reaction, with disastrous results.
The ‘Chamber of Horrors’
Franklin D. Roosevelt began his first term of office in 1933 and part of the new administration’ reform agenda was to revise the 1906 Food and Drugs Act. A draft of proposed changes was submitted in June of that year but it took five years for the legislation to be passed. During the five-year delay, the Food & Drug Administration (FDA) of the U.S. Department of Agriculture engaged in an extensive publicity campaign in support of the new legislation.
One of the FDA’s educational strategies was to set up public exhibits at agricultural fairs (Kay, 2001, p. 457). In 1933, the FDA installed a display at the Chicago World’s Fair specifically designed to highlight deficiencies in the 1906 Act. The stand included ‘before and after’ photographs of the woman blinded by Lash Lure under the heading “Cosmetics are not subject to the present law. Some are dangerous.”
At the end of the World’s Fair the FDA returned the exhibit to Washington, DC where Eleanor Roosevelt, along with a number of prominent Washington women, were invited to see it. The media coverage that followed the visit to the FDA by the First Lady meant that the exhibit attracted national attention. In an article entitled ‘Eyes and Dyes’, TIME magazine noted:
In the Department of Agriculture’s “chamber of horrors” last month Mrs. Franklin D. Roosevelt discovered two photographs, pressed them to her breast crying, “I cannot bear to look at them”. The photographs were of a woman who had got some “Lash-Lure,” an eyebrow & eyelash dye, in her eyes.
(TIME, Dec 4, 1933)
The publicity surrounding Eleanor’s visit to the FDA’s ‘Chamber of Horrors’ led to the Paramount newsreel of October 1933 and the drop in mascara sales. Mascara producers, such as the Kurlash Company and Maybelline, protesting strongly to Paramount, the FDA and others about the damage done to the credibility of their products. Maybelline was particularly concerned as they used the phrase “eyelash beautifier” liberally in their advertising. In a letter to TIME magazine, Tom Lyle Williams, the founder of Maybelline, made the case clearly.
As the manufacturers of the largest-selling, best-known mascara in the world, Maybelline, an absolutely harmless, non-smarting eyelash darkener that contains no dye or aniline derivative, we have suffered untold damage to our old established business by the ambiguous publicity given out concerning the Tugwell Bill. In a recent issue of the Paramount Newsreel, Professor Tugwell told a truly appalling tale of injuries caused by a poisonous preparation, but neglected to give its name as “Lash-Lure” or to state that it was a dye, merely calling it an eyelash “beautifier,” and concluding his speech with the dreadful remark, “This is the kind of stuff you women use on your eyelashes!”
You can imagine how utterly damaging this was to us, inasmuch as the phrase “eyelash beautifier” is practically synonymous with our trade name Maybelline, due to our product being the most extensively advertised mascara on the market for the past 16 years. …
In reply to our protest wired Professor Tugwell and President Roosevelt, Tugwell wrote that he and the department regretted any damage caused us, and that a press dispatch had been immediately issued revealing the name and nature of the offending product, also stating in reference to our product Maybelline that “…we have never heard of any reports of injury caused by it.” Considering that Maybelline has been used consistently every day by millions of women in all parts of the world for over 16 years, this last statement was indeed complete exoneration of our product. However, it did not reach the millions who heard the newsreel speech, therefore, you will realize with what relief and satisfaction we read your discriminating account of the Tugwell propaganda (TIME, Dec. 4), knowing that the real facts in the case would reach your many intelligent readers.
Again thanking you for the inestimable service you have rendered us and all other reputable manufacturers by your careful and authoritative presentation of the news, we are
THOMAS L. WILLIAMS
Maybelline Co. Chicago, Ill.(TIME, Jan 1, 1934)
Williams also took more drastic action and replaced the phrase ‘eyelash beautifier’ with ‘mascara’ in its advertising.
See also Maybelline
Paraphenylenediamine
The German chemist August Wilhelm von Hofmann [1818-1862] had discovered p-phenylenediamines (PPDs) in 1854 but it was not until 1883 that a patent was granted to P. Monnet et Cie of Paris for the use of paraphenylenediamine as a dye. The dye was used in the fur trade but by the mid 1890s it was also employed by hairdressers in Europe and in North and South America to dye human hair (Wall, 1957, pp. 497-498). PPD is an oxidation hair dye that requires an agent to bring out the colour. Initially the dye and other agents were mixed by hand in the salon but, in 1910, Gaston Boudou (Monsieur Emile), a well-known hairdresser with salons in Paris and London, produced the first commercially available PPD hair dye under the trade name Inecto.
For the dyeing of living hair, the earliest method of applying p-Phenylenediamine was in two steps: (a) wetting the hair with a 1 to 3% solution of the dye, made alkaline with caustic soda, sodium carbonate, or ammonia water; (b) applying oxidizing solution to bring out the colour. Many familiar oxidizing agents were used: ferric chloride, potassium dichromate and permanganate, hydrogen peroxide. It was soon found, however, that the best results were obtained with ammonia as the alkalizer and hydrogen peroxide as the developer, and by mixing the two solutions immediately before use. p-Phenylenediamine became popular as a hair dye because it could produce a good, natural-looking, lustrous black, in contrast to the dull, harsh, dusty, greenish or iridescent blacks produced by the prevalent metallic dyes. o-Aminophenol and p-Aminophenol (or their hydrochlorides) were soon adopted for, respectively, orange-red and medium brown shades; and it was found that by varying concentrations and mixing these basic colors several other shades could be produced.
(Wall, 1957, pp. 497-498)
Skin reactions
Reports of skin reactions to PPD and other oxidative hair dyes appeared soon after they started to be used. They became so common that a number of European countries placed restrictions on the sale and use of PPD but most stopped short of a complete ban.
The reactions to PPD appeared to be ‘idiosyncratic’, with only some individuals having a susceptibility or predisposition to ‘hair dye poisoning’. Today we recognise the ‘poisoning’ as an allergic reaction but in the early part of the twentieth century studies into allergies were still in their infancy. Paraphenylenediamine is now considered to be a very potent contact allergen.
French doctors were among the first to recognise the ‘idiosyncratic’ relationship and two skin specialists, Drs. Sabourand and Rousseau introduced a skin test that could be used by hairdressers to see if their clients were ‘susceptible’ to PPD.
Eyes are particularly prone to a severe reaction if the person is allergic. The skin surrounding eyes has the thinnest stratum corneum and the moist conjunctiva of the eye readily absorbs most chemicals it contacts. Individuals who are allergic to PPD quickly develop acute dermatitis and conjunctivitis following exposure to the allergen once primed to react. This makes the use of PPD in eyelash dyes more serious than when used on head hair.
Reactions to PPD can still be serious but are not as dangerous as they were in the past. There are modern treatments for allergic reactions that were unavailable in the 1930s and also, should the eye or surrounding tissue become infected with Golden Staph, antibiotics can be used to treat the infection. These had yet to be developed and only became widely available during the Second World War.
The 1938 Food, Drug and Cosmetics Act
Although the FDA used the Lash Lure scandal in its campaign for reform of the 1906 Act another incident was required for the new Food, Drug and Cosmetic Act (FD&CA) to get passed into law. In 1938, about 100 people, many of them children, died after ingesting an elixir of sulfanilamide that used diethylene glycol as its base. This tragedy and the resultant adverse public reaction finally kicked the U.S. Congress into action and the FD&CA was passed later that year.
The 1938 Act gave the FDA new seizure powers and Lash Lure was the first product it confiscated after the Act was passed. The FDA also issued a statement to eyebrow and eyelash dye manufacturers that it considered the use of paraphenylenediamine and paratolutylenediamine to be product adulterations that would result in prosecution under Section 601 (a) of the Act. This effectively banned their use in the United States.
PPDs in eyelash dyes today
The U.S. ban on the use of PPDs in eyelash dyes is not typical and in most parts of the world eyelash dyes can be found with PPDs in them. Combinal, for example, one of the world’s top-selling eyelash dyes, contains a 2.38% concentration of PPD. It has warnings on its labels including: ‘Can cause an allergic reaction’, ‘Contains phenylenediamines’, ‘For professional use’ and ‘Use suitable gloves’ (Teixeira, De Wachter, Ronsyn, & Goossens, 2006, p. 93) but these warnings rarely appear on websites that advertise the product. So, although these products are designed for salon use only, it seems likely that increased Internet sales may make allergic reactions to PPD more common.
First Posted: 29th November 2010
Last Update: 29th July 2022
Sources
Foan, G. A., & Wolters, N. E. B. (Eds.). (1958). The art and craft of hairdressing: The standard and complete guide to the technique of modern hairdressing, manicure, massage and beauty culture (4th ed.). London: New Era Publishing.
Kay, G. (2001). Healthy Public Relations: The FDA’s 1930s Legislative Campaign. Bulletin of the History of Medicine, 75(3), 446-487.
Kay, G. (2005). Dying to be beautiful: The fight for safe cosmetics. Columbus, Ohio: Ohio State University Press.
Phillips, M. C. (1934). Skin deep: The truth about beauty aids. New York: Garden City Publishing.
Teixeira, M., De Wachter, L., Ronsyn, E, & Goossens, A, (2006). Contact allergy to para-phenylenediamine in a permanent eyelash dye. Contact Dermatitis, 55, 92-94.
TIME Magazine.
Wall, F. E. (1957). Bleaches, hair colorings, and dye removers. In E. Sagarin, (Ed.) Cosmetics: Science and technology (pp. 479-530). New York: Interscience Publishers, Inc.

Lash Lure box with individual packets of dye.
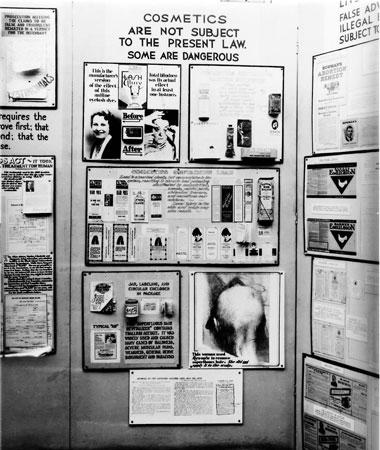
1933 Part of the FDA display for the Chicago World’s Fair. This section, displayed under the prominent heading “Cosmetics are not subject to the present law. Some are dangerous” included a section on reactions to Lash Lure.

Closer view of the FDA display highlighting the dangers of Lash Lure that included before and after photographs of the woman who was blinded after using Lash Lure.

1930 V Spiro Coloura eyelash and eyebrow dye.

1932 Ey-Teb, Inc. Ey-Tec eyelash and eyebrow dye.

1934 Maybelline. The company is stressing that its product is not a dye and is completely safe.
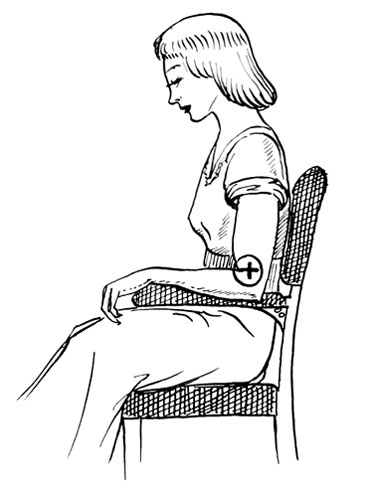
Patch tests for eyelash dyes are usually done behind the ear. The first exposure to an allergen will sensitise an individual but will not provoke a reaction. So, a negative patch does not conclusively mean a person is not allergic to the chemical substance. The description below is for a patch test to hair dye from the 1950s.
Sabouraud-Rousseau Test for Hair Dyes
“The test for liquid dyes is carried out as follows. Pour a few drops of the bottles marked “A” and “B” and mix them together in a small vessel. Then, with an orange stick or match stick around the end of which has been wrapped a small piece of cotton wool, make a cross on the arm of the prospective client. After a few minutes, when the dye has dried on the skin, apply a little collodion with a camel hair brush (new-skin is equally suitable) covering the whole of the cross and allow to dry, thereby forming a protective film. Leave this undisturbed for not less than twenty-four hours, when the collodion or new-skin should be peeled off and the patch washed with soap and water. If no irritation has been experienced, and their is no redness or inflammation, then the client’s skin is free from predisposition, and the full dying process can be undertaken.
For cream dyes mix a small dab of cream in a glass or porcelain dish with a few drops of hydrogen peroxide and apply as explained in regard to liquid para dyes.
Needless to say, the above test must not be regarded as absolutely infallible, but, nevertheless, it is the most reliable and practical test yet devised for determining the presence or absence of predisposition.” (Foan, 1958, p. 365-366)

Combinal, one of many eyelash dyes currently on the market that contain PPD. If proper protocols are followed it should not prove to be a major problem.
